Top 10 things to review this end of year in relation to product governance.
A recent survey found that more than half of the product managers indicate that their product management process was ineffective for ongoing products (Product Management Trends & Benchmarking Report 2019). Most of the product managers surveyed also expect product governance to be driven by data next year.
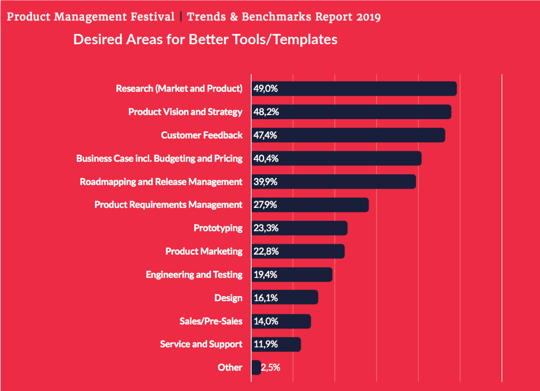
Don’t settle for an unproductive and old-fashioned approach to product lifecycle management. Effective product governance has a positive impact on collaboration, productivity and customer trust. A do-it-yourself checklist is a great place to start. To refresh your product governance, we have created this end of year checklist:
1. Is customer trust a core metric of your product management?
Does your product management framework include monitoring customer trust as a core goal of your product management purpose and strategic direction? Are you actively setting triggers on areas like customer complaints and cancellations?
2. Do you review the domains of product health as part of annual review process?
Have you defined the product manager role as accountable for product oversight and the single point for the end-to-end view of product health? Do you undertake initial and annual product reviews across the 5D's of product health?
3. Do you have a consistent product monitoring cadence?
Do you have a consistent product auditing cadence? The most effective cycle is a consistent one. We suggest the product manager monitors and reviews product health with a monthly, quarterly and an end of year schedule.
4. Are you setting action plans to address assessed risk?
Verify that you are completing, documenting and communicating regular risk profiling for each product and setting action plans to address those risks.
5. Are you communicating product health effectively throughout the organisation?
Do you consistently share product strategy updates with your Board and senior executives that report on product health, including operational financial and non-financial metrics.

6. Are regulatory requirements being met?
Verify that customer and regulatory requirements are determined and understood for each product. A review of new or changed conditions should be part of the product monitoring and review schedule.
7. Do you have a risk notification and escalation process?
Have you defined your product risk appetite, target market persona and trigger events for each product? Do you ensure that critical challenges and risks go through a notification and escalation process?
8. Do you regularly review your distribution channels
Ensure distribution channels are appropriate for target market and monitor your products sold in that channel. Establish 90-day product health reviews.
9. Are you getting input from all areas of the business?
Governance should include a virtual team with representation across the business to collaborate around the product health.
10. Do you have the information you need when the regulator calls?
You should have a single location for product lifecycle information that is easily accessible. Your product lifecycle history should also be updated consistently.
Getting product lifecycle management right is critical to success. If you want to find out how Skyjed can help improve compliance and strengthen your product with a modern product governance platform that unifies design, monitoring, review and improvement with no code automation, feel free to get in touch.
Check out the Skyjed blog for more insights and tips on product lifecycle management.
